|
It suggests that there is more involved than simply overcoming a larger ionic charge. The third IE, however, is over five times the previous one. The second IE is twice the first, which is not a surprise: the first IE involves removing an electron from a neutral atom, while the second one involves removing an electron from a positive ion. Mg(g) \longrightarrow Mg +(g) + e − IE 1 = 738 kJ/mol For example, the following are the first three IEs for Mg, whose electron configuration is 1 s 22 s 22 p 63 s 2: However, IE takes a large jump when a successive ionization goes down into a new shell. This is because more than one IE can be defined by removing successive electrons (if the atom has them to begin with):Ī(g) \longrightarrow A +(g) + e − IE 1Ī +(g) \longrightarrow A 2+(g) + e − IE 2Ī 2+(g) \longrightarrow A 3+(g) + e − IE 3Įach successive IE is larger than the previous because an electron is being removed from an atom with a progressively larger positive charge. IE also shows an interesting trend within a given atom. Again, the trend isn’t absolute, but the general trends going across and down the periodic table should be obvious.įigure 2. 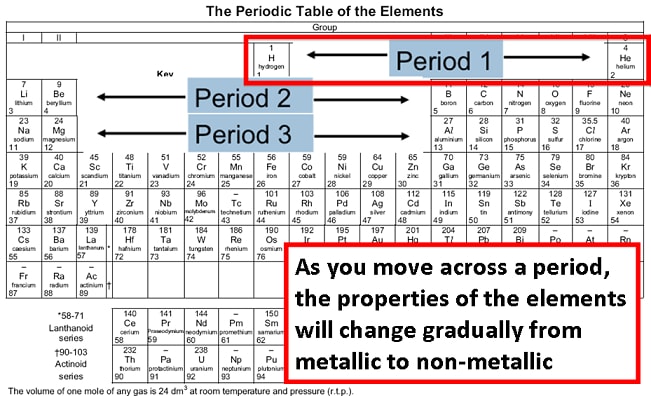
However, as you go across the periodic table and the electrons get drawn closer in, it takes more energy to remove an electron as a result, IE increases.įigure 2 “Ionization Energy on the Periodic Table” shows values of IE versus position on the periodic table. As you go down the periodic table, it becomes easier to remove an electron from an atom (i.e., IE decreases) because the valence electron is farther away from the nucleus. It is always positive because the removal of an electron always requires that energy be put in (i.e., it is endothermic). IE is usually expressed in kJ/mol of atoms. Ionization energy (IE) is the amount of energy required to remove an electron from an atom in the gas phase:Ī(g) \longrightarrow A +(g) + e − ΔH ≡ IE Thus, we expect that as you go from left to right along each period, ATOMIC SIZE DECREASES. These protons serve to pull the electrons closer to the nucleus. 
0 Comments
Leave a Reply. |
Details
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
